Pineamin® is the first injection medicinal product to treat menopausal syndrome with innovative PPG (Polypeptides of Pineal Gland) class effect and Anti-age effect.
Pineamin® is prescribed to treat menopausal symptoms such as hot flashes, sweating, insomnia, depressions, irritability. Such symptoms may occur both in perimenopause (menopausal transition and menopause) and postmenopause.
PPG peptides Peniamin contains stimulate secretion of endogenic melatonin by the pineal gland that regulates pulse secretion of gonadotropin-releasing hormone, FSH and LH.
Endogenic melatonin has a positive Anti-age effect on age-related changes in skin.
Due to the central mechanism of action and injection form Pineamin® has no proliferative effect on endometrium and no negative effect on the blood-clotting system and liver function.
Within 180 days in the double blind randomized placebo-controlled study Pineamin® was not different from placebo in terms of safety regarding increase in the thickness of endometrium, cervix cytology, effect on hemostasis,liver function and lipid profile.
The drug should be administered within short 10-day injection courses. Effectiveness of Pineamin remains for several months after each course of injections. Repeated treatment is prescribed in 3-6 months, if necessary.
This drug is a prescribed medicine. It can be dispensed on a doctor's prescription only. There are contraindications. Please read the patient information leaflet. Do not use the medicinal agent if it was not prescribed by your treating physician. Always follow the patient information leaflet and your doctor’s recommendations.
Official website – www.pineamin.ru
Anatomic proximity of hypothalamus structures responsible for reproductive function and the structures relating to regulation of vasomotor reactions underlies the symptoms of menopausal syndrome such as hot flashes, sweating and other symptoms.
Pineamin effect is based on regulation of the central hypothalamus-hypophysis link n terms of age-related estrogen deficit.
The level and the pulse rhythm of gonadotropin-releasing hormone secretion in hypothalamus is defined by the interaction (balance) of between two groups of secreting neurons: kiss-stimulating neurons gonadotropin-inhibitory (GnIH) neurons. During age-related changes estrogen deficit causes disruption of the pulse rhythm gonadotropin-releasing hormone secretion level resulting from the dominance effect of kiss-stimulating hormones as a response to reduced ovarian production of estrogens.
Gonadotropin-inhibitory neurons are stimulated by endogenic melatonin with the pineal gland located in the brain area adjacent to hypothalamus being its main source of secretion.
Pineamin® containing PPG (PolypeptidesofPinealGland) peptides recovers functional density of the pineal gland, stimulates endogenic melatonin secretion by the pineal gland and thus recovers the balance of interaction between kiss-stimulating neurons and gonadotropin-inhibitory neurons and gets back the right pulse rhythm and secretion level of gonadotropin-releasing hormone, FSH and LH.
Endogenic melatonin has a positive Anti-age effect on age-related changes in skin by strengthening the epidermal barrier and maintaining skin turgor.
TV seminar by Kasyan V. N., Candidate of Medical Sciences, obstetrician and gynecologist, assistant professor at the Department of Reproductive Medicine and Surgery in A. I. Yevdokimov Moscow State University of Medicine and Dentistry on the following topic: Neurohumoral regulation of the reproductive system. Place of PPG in correction of menopausal complaints. Available at http://pineamin.ru/video/video.html.
Group name: pineal gland polypeptides of the cattle.
Dosage form: lyophilisate for preparation of solution for intramuscular injections.
Composition
Pineamin medicinal product®, lyophilisate for preparation of solution for intramuscular injections 10 mg, is a complex of water-soluble pineal gland polypeptide fractions of the cattle not older than 12 months separated from Pineamin® dried extract (GEROPHARM LTD, Russian), containing glycine as a stabilizer (Ajinomoto Co. Inc., Japan; Panreac, Spain) in ratio 1: 2.
Description
White or white and yellow powder or porous mass.
Pharmacotherapeutic group: anti-menopausal medicinal product.
ATC code: G02CX.
Pharmacodynamics
The medicinal product optimizes the relationship between the pineal gland and hypothalamus, normalizes function of the hypophysis front lobe and the balance of gonadotropic hormones.
During the double blind randomized placebo-controlled study of the medicinal product in the population of women during postmenopause fair less marked symptoms of menopausal disorders were noted according to Kupperman Index caused by positive effect of the medicinal product on neurovisceral manifestations of menopausal syndrome.
Earlier in pre-clinical studies with participation of experimental animals during long-term (30 and 90 days) intramuscular administration of PINEAMIN negative effect on major systems (nervous, cardio-vascular, haematogenous, urinary and respiratory) and also on the metabolism and general body condition were not identified.
Indications for use
Neurovisceral disorders in menopausal syndrome in women in case of contradictions to hormone replacement therapy (HRT) or in case of refusal from it.
Contradictions
Hypersensitivity or intolerance of any component of the medicinal product, age before 18, pregnancy and breastfeeding, metrorrhagia (spotting of unclear genesis). Pre-cancer and malignant diseases including estrogen-dependent tumors of reproductive organs and breasts.
Cautiously
The medicinal product has no effect on concentration of sex hormones in plasma. However, it should be used cautiously in uterine leiomyoma and endometriosis .
Use in pregnancy and breastfeeding:
the medicinal product should not be used during pregnancy and breastfeeding.
Directions for use and dosage
The medicinal product should be administered intramuscularly.
Before injecting dissolve the contents of the vial in 1–2 ml of 0.5 % procaine solution (novocaine), water for injections or 0.9 % sodium chloride solution and administer one dose of 10 mg once a day within 10 days.
Repeated treatment is prescribed in 3-6 months, if necessary.
If you missed an injections, you should not administer double dose but make the next injections as scheduled.
Side effect
Allergic reactions are possible in case of individual intolerance to any component of the medication.
In the clinical study the following individual unwanted reactions were observed MedDra classification):
| Class according to MedDra | Unwanted reaction | Frequency (WHO classification) |
| Genital-related and breast-related disorders | spotting | Not frequent (≥ 1 %, but < 10 % of prescriptions) |
| Laboratory and instrumental data | increased concentration of blood estradiol | Not frequent (≥ 1 %, but < 10 % of prescriptions) |
| Skin-related and subcutaneous-related disorders | infiltrate in the spot of intramuscular injection | Rare (≥ 0.01 %, but < 0.1 % of prescriptions) |
If mentioned side effects occur, consult a doctor.
If any of the mentioned side effects are getting worse or if any other not mentioned side effects occur, consult a physician.
Overdose:
no overdose cases are described.
The symptoms of overdose may include spotting, increased concentration of blood estradiol. In these cases the drug withdrawal and symptomatic treatment are necessary.
Interaction with other medicinal agents
Interaction with other medicinal agents has not been identified.
Inconsistence
PINEAMIN® solution should not be mixed with other solutions.
Special warnings
PINEAMIN® can be used only when prescribed by a doctor!
To prevent possible individual hypersensitivity to the drug components, trial injection is recommended before therapy.
Do not keep and use the vial containing the drug solution after expiry date.
Special precautions when destroying unused drug are not needed.
Effects on ability to drive and use machines
The effect of the medicinal product on performance of potentially dangerous activities demanding enhanced emphasis and focus and fast psychomotor reactions has not been identified.
Dosage form
Lyophilisate for preparation of solution for intramuscular injections, 10 mg.
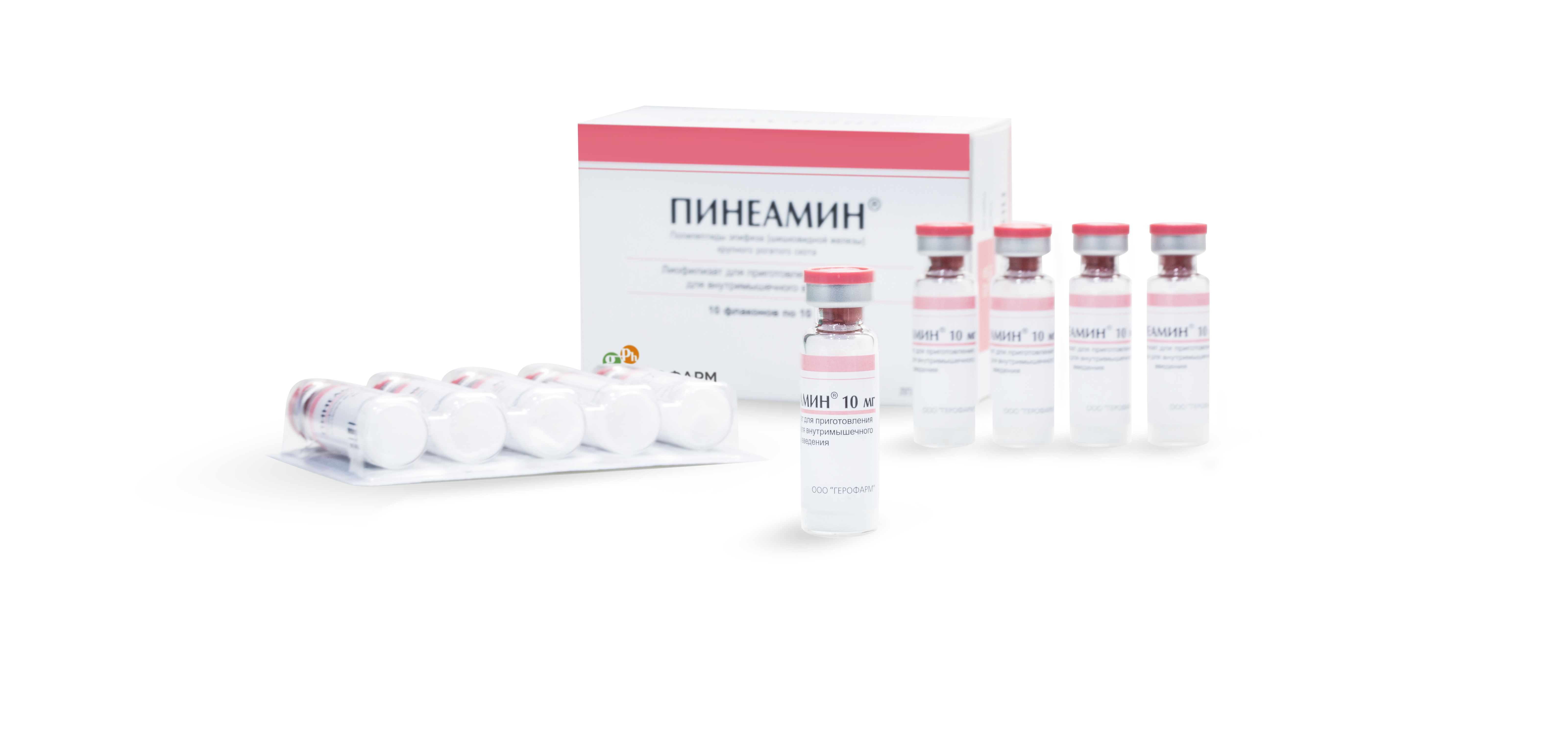
5-ml colorless glass vials with aluminum caps with pink or orange tear-off plastic cover having an embossed sign: GEROPHARM. 5 vials in a contour cellular pack made of PVC film and aluminum foil. One or two contoured packs with instructions for use are placed in a carton box.
Storage conditions
Protected from light at 2–20 °C.
Keep out of reach of children.
Shelf-life
3 years.
Dispensing conditions
By prescription.
Registration number: LP-003202
The firm accepting claims
GEROPHARM LTD, Russia
B-11, Degtyarny per., Saint Petersburg, 191144
Phone: (812) 703-79-75 (MLT)
Fax: (812) 703-79-76
Toll-free number: 8-800-333-4376 (free call across the Russian Federation)
Pineamin® is a medicinal product based on lyophilisate of pineal gland polypeptide fractions (PPG – Polypeptides of Pineal Gland) for preparation of the solution for intramuscular injections. Glycine amino-acid supplied for Pineamin production from Japan or Spain is used as a stabilizer-antioxidant.
Pineamin® is supplied in vials with 10 of them places in a carton box (two contour cells with 5 vials each).
Package solutions are intended to provide for convenience, safety and control of Pineamin use.
The package has several types of informational indications.
The informational indications are visual and tactile – for patients with poor eyesight.
Color indication of Pineamin looks like light-pink strips on the package and a plastic temper-evident cover of the same color.
Tactile indication of Pineamin is given as a convex indication (Braille).
Safety and protection of a patient against falsification has several levels:
Control of the first opening of the package in the form of a valve with a perforating edge.
Control of the first opening of the vial in the form of a color plastic cover.
Free edge of blister foil provides for convenient removal of the vial.
All the surfaces of a blister with Pineamin vials are rounded without sharp edges. This technological peculiarity helps avoid cutting and piercing fingers when removing the vials.